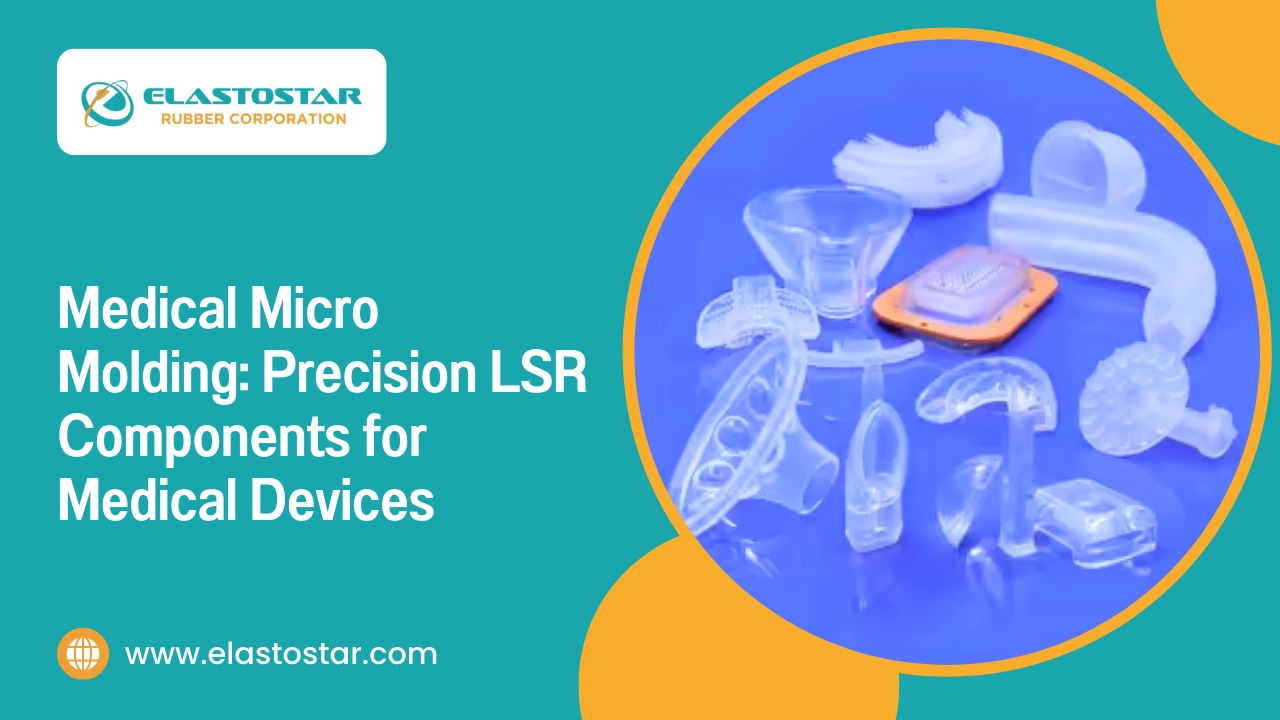
Medical devices are becoming smaller, more complex, and more performance-critical, driving demand for manufacturing processes that deliver extreme precision at the microscale. Medical micro molding addresses this need by enabling the production of sub-gram extruded silicone components with tight tolerances, consistent geometry, and repeatable quality.
When combined with liquid silicone rubber, micro molding provides the biocompatibility, flexibility, and durability required for regulated healthcare applications. From catheter components to miniature valves and seals, medical LSR molding plays a vital role in modern device design.
This guide explains the process, precision requirements, and validation considerations behind producing reliable micro-molded silicone components for medical devices.
Key Takeaways
- Medical micro molding enables the production of sub-gram, high-precision silicone components.
- Liquid silicone rubber offers flexibility, durability, and biocompatibility at the microscale.
- Tight tolerances are critical to ensure proper fit, sealing, and device performance.
- Medical micro-molded parts must meet USP Class VI and ISO 10993 requirements.
- Validation and testing ensure consistent quality and regulatory compliance.
- Micro molding enables the production of advanced medical devices, including catheters, valves, and seals.
- Precision tooling and controlled processes are essential for repeatable results.
Table of Contents
What is Medical Micro Molding?

Medical micro molding is a specialized manufacturing process used to produce very small, high-precision components, often weighing less than 1 g and featuring micro-scale geometries.
In medical micro molding, this process becomes essential when conventional injection molding cannot meet the required accuracy, consistency, or part integrity.
Key characteristics of medical micro molding include.
- Production of sub-gram and micro-scale components.
- Extremely small shot sizes with tight process control.
- Complex geometries with fine details and thin sections.
- High repeatability for regulated medical applications.
At this scale, even slight variations in material flow, temperature, or tooling can impact performance. For this reason, medical micro molding relies on precise tooling, controlled injection parameters, and stable material behavior.
Liquid silicone rubber is widely used for its flexibility, durability, and biocompatibility, as outlined in our silicone material overview.
At Elastostar Rubber Corporation, we apply these micro-molding principles to produce miniature medical components where dimensional accuracy and consistency are critical.
Also Read– The Step-by-Step Process of Custom Silicone Rubber Molding
LSR Micro Molding Process Specifications

Medical LSR micro molding requires tight control at every stage to produce consistent, high-precision microscale parts.
Liquid silicone rubber is well-suited for micro injection molding because it flows easily, fills fine details, and cures into a stable, elastic material.
Key process specifications in medical LSR micro molding include.
- Precise shot size control to manage extremely small material volumes.
- High-accuracy tooling designed to fill thin walls and micro features.
- Controlled injection pressure and speed to avoid flash or short shots.
- Stable curing conditions to maintain dimensional consistency.
- Clean manufacturing environments to support medical device requirements.
Process repeatability is critical in medical applications, as even minor variation can affect fit, sealing, or device function.
This is why micro molding operations rely on documented controls, in-process monitoring, and inspection practices similar to those outlined in Elastostar’s quality control approach.
By combining controlled LSR processing with precision tooling, medical micro molding supports reliable production of miniature silicone components for regulated medical devices.
Also Read–3 Types of Silicone Rubber Molding Processes
Tight Tolerance Capabilities (±0.001 Inches)

Medical micro molding demands extremely tight dimensional control, as even small deviations can affect device performance, sealing integrity, or patient safety.
In medical micro molding, tolerances as tight as ±0.001 inches are often required to ensure precise fit and repeatable function.
Why tight tolerances matter in micro-molded medical parts.
- Ensure proper sealing and flow control in miniature components.
- Maintain consistent fit within complex medical assemblies.
- Reduce the risk of leakage, misalignment, or device malfunction.
- Support interchangeability across high-volume production runs.
Achieving this level of precision requires more than advanced tooling. It depends on controlled material behavior, stable molding parameters, and strict dimensional verification.
We apply established tolerance controls to ensure consistency across production, in accordance with industry-recognized practices for precision elastomer components.
By maintaining tight tolerances at the microscale, the ISR medical device ensures reliable performance in applications where accuracy cannot be compromised.
Biocompatibility Requirements (USP Class VI, ISO 10993)

Biocompatibility is a critical requirement for any component used in medical devices & pharmaceutical industry, especially those that contact fluids, tissue, or sensitive internal environments.
In precision medical silicone, material selection must meet strict regulatory and safety standards to ensure patient protection and device reliability.
Key biocompatibility requirements for micro-molded medical parts include.
- USP Class VI compliance, which evaluates biological response under controlled testing.
- ISO 10993 standards, covering cytotoxicity, sensitization, and irritation.
- Material traceability and controlled formulation consistency.
- Documentation to support regulatory submissions and audits.
Liquid silicone rubber is widely used in medical applications because it meets biocompatibility requirements while maintaining flexibility and durability.
We support medical programs with materials and processes aligned with certified quality and compliance practices, as outlined in our certifications and quality documentation.
Meeting biocompatibility standards at the micro scale ensures that precision LSR components perform safely and consistently throughout the medical device lifecycle
Also Read– Is Medical Grade Silicone FDA Approved
What are the Medical Applications of Micro-Molded LSR Components

Medical micro molding enables the production of miniature silicone components used in precision-critical areas of medical devices.
At the microscale, consistency, flexibility, and biocompatibility are essential to ensure reliable device performance and patient safety.
Common medical applications include.
- Catheter tips and distal components, where smooth geometry and dimensional accuracy support controlled insertion and fluid flow.
- Micro check valves are used to regulate fluid movement in diagnostic and therapeutic devices.
- Surgical seals and miniature gaskets are designed to maintain sealing integrity in compact assemblies.
- Fluid-contact components, where material purity and repeatability are required.
Liquid silicone rubber is well-suited for these applications due to its flexibility, stability, and compliance with medical regulatory requirements.
Micro-molded LSR components are often produced as precision-molded silicone parts that integrate seamlessly into complex medical assemblies.
By supporting these applications, medical micro molding plays a key role in advancing compact, high-performance medical device designs.
Validation and Testing Requirements
In micro injection molding, validation and testing ensure that precision components meet dimensional, functional, and regulatory requirements before they are approved for use in medical devices.
Because micro-molded parts often serve critical roles, verification must be thorough and repeatable.
Key validation and testing steps for medical LSR molding include.
- Dimensional inspection using high-resolution metrology to verify micro-scale features produced through micro-injection molding.
- Process validation (IQ, OQ, PQ) to confirm stable and repeatable manufacturing performance.
- Material verification to ensure LSR compounds meet medical-grade and biocompatibility requirements.
- Functional testing to validate sealing, flow control, or mechanical behavior of biocompatible micro parts.
- Documentation and traceability to support regulatory review and quality audits.
By combining controlled processes with structured validation, medical micro molding delivers consistent, compliant, and reliable LSR components for medical devices.
Why Choose Elastostar Rubber Corporation for Medical Micro Molding?

Producing micro-scale components for medical devices requires more than precision tooling. It demands material expertise, regulatory awareness, and consistent process control.
As a custom silicone products manufacturer, we support medical micro molding programs with a focus on quality, compliance, and repeatability.
Why medical device manufacturers work with us.
- We specialize in medical LSR molding of precision silicone components for regulated applications.
- Our materials meet USP Class VI requirements, supporting biocompatible micro parts for medical use.
- We offer tight-tolerance manufacturing for micro-injection molding and complex geometries.
- We support design assistance, prototyping, and production runs, helping transition concepts into manufacturable parts.
- Our processes are built around documented quality control and validation practices.
- All manufacturing is proudly made in the USA, ensuring supply chain reliability and traceability.
By combining precision manufacturing with medical-grade materials and validated processes, we help ensure micro-molded silicone components perform reliably throughout the medical device lifecycle.
Recommended Reads
- Why Medical Grade Silicone Tubing Is the Best Choice for Healthcare Tubing
- What Makes Silicone Rubber Popular for Use in the Healthcare Industry?
- Silicone Rubber for Medical Equipment Manufacturers
Conclusion
Medical devices continue to demand smaller, more precise, and more reliable components across critical healthcare applications. Medical micro molding enables the production of sub-gram silicone parts that meet tight tolerance, biocompatibility, and functional requirements.
When combined with liquid silicone rubber, micro molding supports consistency, flexibility, and long-term stability in demanding environments. Successful outcomes depend on precision tooling, controlled processing, and thorough validation aligned with medical regulations.
We support medical LSR molding programs from design through production using validated quality systems and manufacturing expertise. If you are developing micro-scale silicone components, contact us to discuss performance goals and compliance needs.
FAQs
Q1. What is medical micro molding?
Medical micro molding is a precision manufacturing process used to produce extremely small, high-accuracy components for medical devices. It is commonly applied to sub-gram parts that require tight tolerances, repeatable geometry, and medical-grade materials.
Q2. How small can LSR micro-molded parts be?
LSR micro-molded parts can weigh under 1 gram and may include features measured in fractions of a millimeter. The exact size depends on part geometry, wall thickness, and tooling design.
Q3. What tolerances are achievable with micro molding?
In medical micro molding, tolerances as tight as ±0.001 inches are achievable. Maintaining these tolerances requires precision tooling, controlled material flow, and stable processing conditions.
Q4. What biocompatibility standards apply to micro-molded parts?
Micro molded medical components typically must meet USP Class VI and ISO 10993 requirements. These standards evaluate biological response, material safety, and suitability for medical use.
Q5. What are typical applications for medical micro molding?
Common applications include catheter tips, micro check valves, surgical seals, fluid-control components, and other miniature parts used in diagnostic, therapeutic, and surgical devices.
Q6. Is LSR better than thermoplastics for micro molding?
LSR is often preferred for medical micro molding because it offers flexibility, thermal stability, and biocompatibility. Thermoplastics are suitable for rigid applications, but LSR performs better for sealing and soft-contact components.
Q7. What is the minimum wall thickness for LSR micro parts?
Minimum wall thickness depends on design and tooling, but LSR micro parts can be molded with very thin, uniform walls. Consistent wall thickness is critical to ensure proper material flow and curing.
Q8. How is USP Class VI testing performed for micro components?
USP Class VI testing evaluates biological response through standardized extraction and exposure methods. For micro components, testing is performed on representative material samples to validate compliance.
Q9. What are the key design considerations for medical micro molding?
Important design factors include uniform wall thickness, adequate draft angles, proper gate placement, and simplified geometry. Early collaboration between design and manufacturing teams improves moldability and yield.
Q10. What is the lead time for micro-molded medical prototypes?
Lead times vary based on complexity, tooling requirements, and validation needs. Prototype timelines are typically shorter when designs are optimized for micro molding, and materials are pre-qualified.